Forrás: Arzneimittel-Forschung (Drug Research)
From the Semmelweis Medical School, Budapest (Hungary), Institute of Pharmacology;
Head: Professor Dr. J. Knoll
By J. Knoll, Z. Mészáros, P. Szentmiklósi, and S. Fürst
During the past few years we have been conducting extensive studies aimed at the recognition of new structures with analgetic activity. In the course of this research work we discovered that some members of the 1,5-diazanaphthalene series exhibited analgetic properties different from those of the hitherto known analgesics (K n o l l 1968; K n o l l et al. 1969).
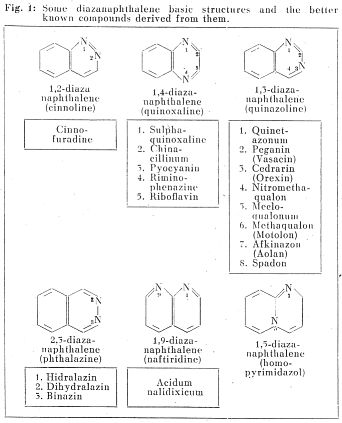
Fig. 1 presents some types of the diazanaphthalene basic structure and some known drugs derived from them. Up to the present the 1,3-diazanaphthalene (quinazoline) structure was found to be the most useful pharmacologically. A small number of drugs pertaining to the cinnolines, phthalazines and naphthyridines are known. No drug containing the 1,5-diazanaphthalene structure, which forms the object of our studies, has been described, as yet. The denomination and designation of the compounds containing this skeleton is not uniform in chemical literature, the numeration seen in Fig. 1. and the trivial name "homopyrimidazol" has been adopted in our studies.
About 250-300 homopyrimidazol derivatives are known in chemical literature, however, reports on pharmacological examination have only been published in connection with the compound substituted on 2-, 3- or 4-position by P r u s s and H i d a l g o (1965) describing its histamine-releasing effect. Derivatives substituted in position 6 have been left out of consideration as - according to the pertaining literature - their synthesis meets with difficulties mainly on account of the labile bicyclic structure.
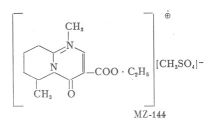
We have elaborated a new method for the synthesis of homopyrimidazols which has the advantage that also derivatives substituted on position 6 can be easily prepared by it (M é s z r o s et al. 1969). The synthesis and pharmacological analysis of 85 comounds helped us to the selection of 1,6-dimethyl-3-carbethoxy-4-oxo-6,7,8,9- tetrahydro - homopyrimidazol methylsulphate (MZ-144) for further detailed studies.
The following is a brief description of some important relations between the structure and activity regarding the homopyrimidazol analgesics.
According to the basic criterion of our evaluations that only compounds producing an analgetic effect at doses below half of the LD50 are considered effective, it was stated that homopyrimidazols bearing -COOC2H5 in position 3 (Table 1) possess advantageous qualities deserving further study. This is clearly proved by the comparison of the effect of compounds MZ-108, MZ-104, and MZ-144 with that of the other derivatives.
Compounds having -COOH on position 3 (e.g. MZ-121, MZ-124, MZ-211) are much less toxic than the esters but inactive.
Derivatives unsubstituted on position 3 display no analgetic activity but their toxicity is greatly increased in comparison to the acid, e.g. the LD50 of MZ-132 is 0.85 mM/kg while that of the corresponding acid (MZ-121) is 9.61 mM/kg.
Saturation of ring A significantly reduces toxicity both in the acids and in the esters derived from them. This correlation clearly appears from the comparison of the analgetic effect of MZ-144 and its hydrated derivative, MZ-121, or when MZ-104, the analgetic ethyl ester, is compared with its hydrated derivative, MZ108. The comparison of MZ-104 and MZ-108 also shows that the pharmacological activity of the compounds almost parallelly increases resp. decreases with toxicity.
It also appears from the data of Table 1 that quaternization of N in ring A produces an effect exactly opposite to hydration because, as it is seen from the comparison of MZ-211 and MZ-121, or MZ-144 and MZ-108, both toxicity and activity are increased following quaternization.
Comparing the esters of derivatives containing a saturated ring A it becomes conspicuous that both from the aspect of activity and toxicity the ethyl ester possesses the most outstanding qualities. Among the derivatives MZ-130, MZ-108, MZ-204, and MZ-136 only the ethyl ester MZ-108 fulfils the criterion of selection for pharmacological activity. The same correlation is found at the comparison of MZ-128 and MZ-104. Concerning activity in oral application, from the derivatives MZ-144, MZ-123, MZ-214, and MZ-215, the ethyl ester, MZ144, possesses the most advantageous qualities.
As unanimously proved by the data of Table 1, analgetic effect is significantly increased by N1-methylation.
On the basis of the data contained in Table 1 the influence of N1-substituted groups on pharmacologic effect was studied. Table 2 contains some data in this connection. Similarly to ring A the saturation of ring B also reduces toxicity to a considerable degree. A good example for this is seen from the comparison of the toxicity data of MZ-168 and MZ-170.
Table 3 presents the activity spectra of some compounds found to exert analgetic activity. On the basis of the LD50/ED50 ratio, evaluation of the pharmacological results in both tests used for screening the analgetic effect and oral activity MZ-144 was found to possess the most advantageous properties.
Not all the compounds were equally active in both tests. E. g. MZ-142 potentiated the effect of morphine both on i.v. and p. os administration to a significant extent, however, when given alone by the oral route, its activity did not reach the required level in the hotplate test. A very significant difference was found between the intravenous and oral effect of other compounds. For instance, MZ-170 displayed favourable qualities in both tests on intravenous administration, however, it was ineffective by the oral route. Besides the compounds listed in the tables more than 50 homopyrimidazols were prepared and tested. In every respect, especially regarding its oral effect, MZ-144 appeared to be the most suited for detailed pharmacological study.
The compound used in our experiments was the methylsulphate of
1,6-dimethyl-3-carbethoxy-4-oxo-6,7,8,9-tetrahydro-homopyrimidazol.
Empirical formula: C14H22N2O2S.
Molecular weight: 362.41.
The compound is a white amorphous powder, readily soluble in water.
Depending on the pH aqueous solution of MZ-144 is either colourless (form A) or coloured
(form B).
Distribution of the two forms in the B r i t t o n - R o b i n s o n
buffer series (standard solution containing 0.04 M acetic acid, 0.4 M phosphoric acid and 0.4 M
boric acid, specified volumes of 0.2 N NaOH to be added to 100 ml of the solution) is shown in
Table 4.
As indicated by our chemical studies (M é s z á r o s
et al.) the base liberated from the salts, including MZ-144, may be characterized by the
following structure:
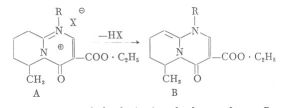
In the case of 6-methyl-substitution the bases of type B are obtained in the form of an oil, no crystallized product could be prepared. Basis of the type B were obtained in crystalline form from salts not bearing a methyl group in position 6. An example to this is the following equation:
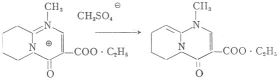
All these data prove that MZ-144 within the organism cannot be considered as a quaternary compound, thus its central activity is not inconsistent with the experiences that true quaternary compounds do not find entrance into the central nervous system.
Among newly synthetized 1,5-diazanaphthalene (homopyrimidazol) derivatives substituted on 1-, 3-, 4- or 6-position analgesics, promising from a practical aspect, were found. Comparing the chemical structure and pharmacologic properties of 85 compounds, 1,6-dimethyl-3-carbethoxy-4-oxo-6,7,8,9-tetrahydro-homopyrimidazol-methylsulphate (MZ-144) was selected as the most suited for detailed pharmacological studies. It was proved that within the organism MZ-144 cannot be considered as a compound containing a quaternary nitrogen.
Acknowledgement
We wish to thank Dr. A. D á v i d and
Dr. G. H o r v á t h for their
valuable help during this study.
Unter den neu entwickelten, an den Stellen 1, 3, 4 und 6 substituierten 1,5-Diazanaphthalin-(Homopyrimidazol)-Derivaten wurden mehrere, in praktischer Hinsicht vielversprechende Verbindungen entdeckt. Die chemische Struktur und die pharmakologischen Eigenschaften von 85 Derivaten wurden miteinander verglichen und auf Grund der Ergebnisse die Verbindung 1,6-Dimethyl-3-carbäthoxy-4-oxo-6,7,8,9-tetrahydro-homopyrimidazol-methylsulphat (MZ-144) zur eingehenden pharmakologischen Untersuchung ausgewählt. Es wurde bewiesen, daá MZ-144 innerhalb des Organismus nicht als eine einen quaternären Stickstoff enthaltende Verbindung zu betrachten ist.
References
For the authors: Prof. D. J, Knoll, Department of Pharmacology,
Medical University, Üllői út 26, Budapest VIII (Hungary)